The Entry-Exit Inspection and Quarantine Bureaus (now operating under the unified name of Customs) primarily undertake the responsibilities of safeguarding national biosafety, economic interests, and public health security, as well as maintaining normal entry-exit order:
- lIn terms of entry-exit goods inspection and quarantine: They are responsible for inspecting import and export commodities to ensure their quality meets national standards, preventing substandard goods from entering the market. Meanwhile, they quarantine entry-exit animals, plants, and their products to prevent the invasion of alien harmful organisms, thus protecting the domestic ecological environment and agricultural security.
- In terms of entry-exit personnel health quarantine: They conduct health checks on entry-exit personnel to prevent the cross-border spread of infectious diseases. They also carry out infectious disease monitoring to promptly detect and control epidemics.
- In terms of certification, accreditation, and laboratory testing: They are responsible for relevant certification and accreditation work to ensure the accuracy and impartiality of inspection and quarantine results. They manage and operate professional laboratories to provide scientific basis for inspection and quarantine.

1、Application of Microscopes in Virus Detection
The Entry-Exit Inspection and Quarantine Bureau has established multiple inspection laboratories, typically including virology laboratories, chemical laboratories, food inspection laboratories, virology laboratories, etc. This article introduces the virology laboratory.
Virology Laboratory
The virology laboratories of the Entry-Exit Inspection and Quarantine Bureau are mainly responsible for monitoring, analyzing, and reporting the epidemic situation of viral diseases, carrying out virus-related research, providing data support for the prevention and control of viral diseases, and offering technical guidance to lower-level laboratories. The functional teams and their corresponding responsibilities are as follows:

(1) Virus Culture Laboratory
It primarily engages in virus isolation, culture, and identification, with capabilities including isolation and identification of influenza virus and poliovirus, neutralizing antibody assay, polio sugar pill titer determination, measles vaccine titer determination, etc. The laboratory is divided into an enterovirus culture area and a respiratory virus culture area, designed as a P2-level biosafety laboratory, and equipped with inverted microscopes, fluorescence microscopes, ultra-low temperature freezers, liquid nitrogen tanks, carbon dioxide incubators, Class II biosafety cabinets, and other equipment.
In the virus culture laboratory, microscopes are mainly used in the following scenarios:
Cell Status Observation
- To observe the morphology and growth status of host cells used for virus culture, such as cell size, shape, and arrangement, and to determine whether cells grow normally, as well as issues like aging or contamination.
- After virus inoculation, regularly observe cytopathic effects to determine whether the virus infects cells and the infection process.
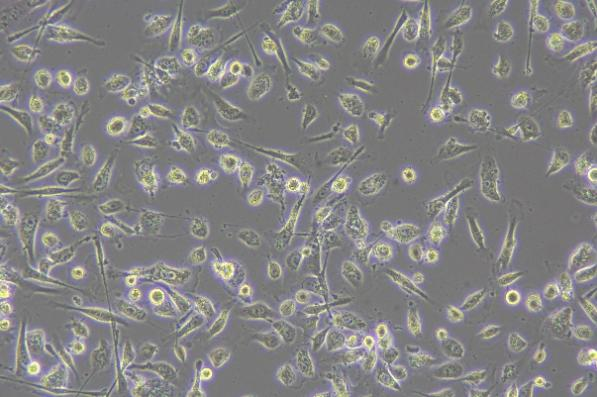
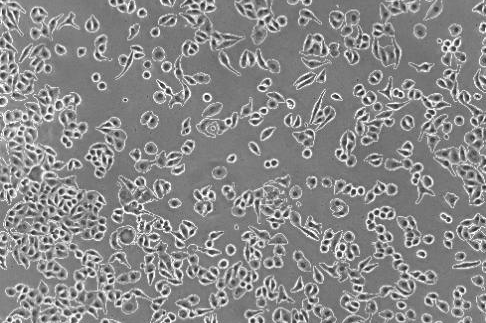
Virus Observation
- Inverted microscopes for virus observation can visualize changes formed by viruses within cells. After infecting cells, some viruses cause structural changes such as cell swelling, deformation, or inclusion body formation. Observing these changes under a microscope allows preliminary judgment of viral growth status.
- lFluorescence microscopes are used to observe fluorescent signals after viruses bind to fluorescently labeled antibodies, determining the location and distribution of viruses within cells for qualitative and localization analysis.
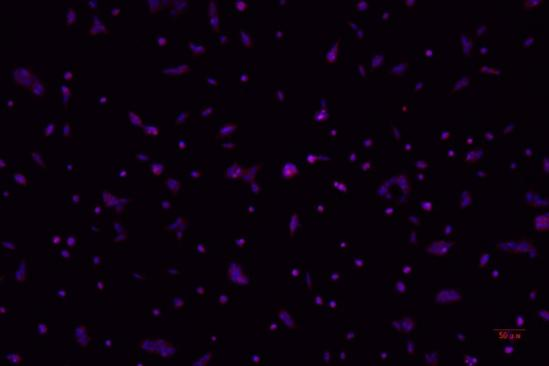
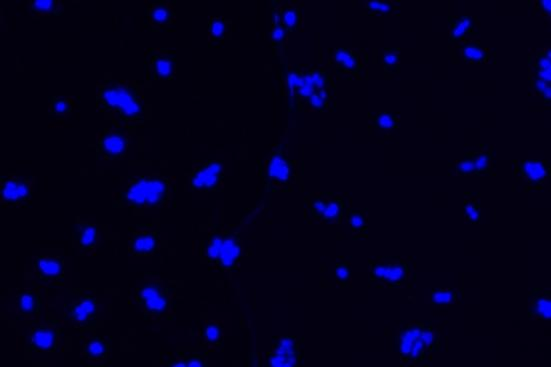
(2) Virus Immunology Laboratory
The Virus Immunology Laboratory undertakes viral immunology detection tasks, capable of conducting tests such as enzyme-linked immunosorbent assay (ELISA), hemagglutination assay (HA), hemagglutination inhibition assay (HI), indirect immunofluorescence assay (IFA), agglutination test, neutralization test, and complement fixation test. Also designed as a P2-level biosafety laboratory, its main equipment includes fluorescence microscopes, ultra-low temperature freezers, ordinary incubators, water baths, Class II biosafety cabinets, microplate readers, plate washers, etc.
In the Virus Immunology Laboratory, microscopes are mainly used in the following scenarios:
Cell status observation
During virus detection experiments, it is necessary to observe the morphology and growth status of host cells used for virus culture, such as whether the cells grow normally, and issues like aging or contamination. Inverted microscopes are used to observe cell size, shape, and arrangement, as well as cytopathic effects after virus inoculation, to determine whether the virus infects cells and the infection process.
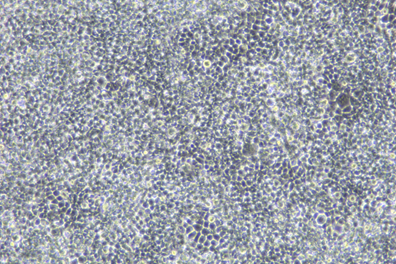
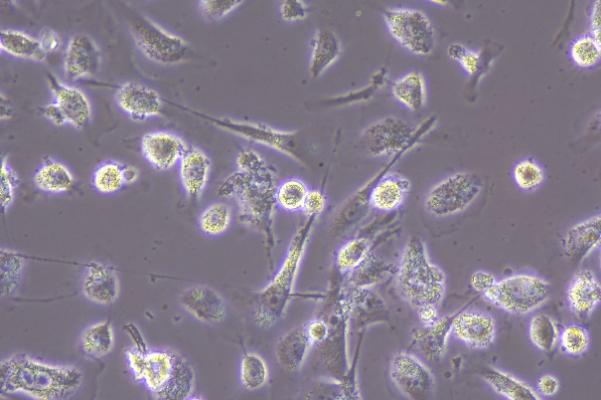
Virus Observation and Localization
Through fluorescence microscopes, using specific fluorescence labeling strategies such as immunofluorescent antibody labeling of viral antigens or nucleic acid probe labeling of viral genomes, viral components can be localized at the subcellular level to determine the location and distribution of viruses within cells, thereby enabling qualitative and localization analysis of viruses.

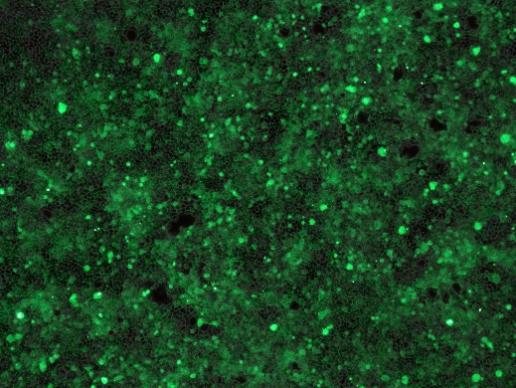
Experimental Result Records
During viral immunology detection experiments, microscopes are regularly used to observe and record the morphological changes of viruses and host cells, providing intuitive image data for the analysis and summarization of experimental results to facilitate subsequent research and analysis.
(3) HIV Laboratory
The HIV Laboratory primarily conducts AIDS screening and confirmation tests, including enzyme-linked immunosorbent assay (ELISA), western blot assay, agglutination test, and gold (selenium) labeled rapid tests. Its development direction includes carrying out nucleic acid analysis, antigen detection, HIV-related immune factor detection, viral load testing, etc. It is equipped with microplate readers, plate washers, automatic western blot instruments, biosafety cabinets, low-temperature freezers, ordinary incubators, automatic high-pressure sterilizers, digital cameras, etc. Flow cytometers are reserved as a development direction.
(4) Biochemical Immunology Laboratory (Hepatitis Room)
The Biochemical Immunology Laboratory (Hepatitis Room) is responsible for immunological and biochemical analysis of hepatitis viruses, divided into ELISA detection area, biochemical analysis area, and time-resolved immunodetection area. Main equipment includes automatic biochemical analyzers, automatic microplate readers, time-resolved detectors, etc.
As shown above, microscopic imaging equipment plays a crucial role in the virology laboratories of entry-exit inspection and quarantine bureaus. With more than 20 years of deep involvement in the field of microscopic imaging, MSHOT has diverse products and rich experience, capable of assisting researchers in their work in the field of virology.
2、MSHOT Recommended Solutions for Virus Detection Field
(1)Recommended Inverted Biological Microscopes and Cameras for Cell Observation
Inverted Biological Microscope MIX60/MIX60-FL + Microscope Cameras MDX10, MSX3, etc.
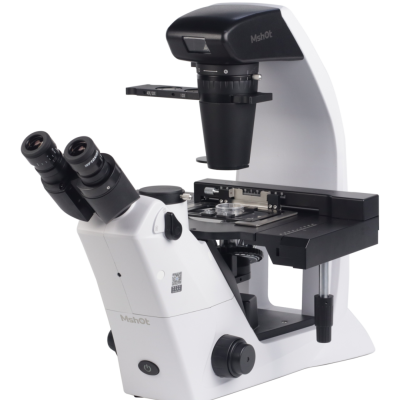
Inverted Biological Microscope MIX60
The MIX60 inverted microscope is a research-grade biological microscope with high-performance imaging, intelligent operation, and clear detail observation. It features a flat field without field curvature, an ultra-long working distance condenser, and supports brightfield and phase-contrast observation. It can be upgraded for fluorescence observation, equipped with an OLED digital display screen and voice broadcasting function to real-time display light intensity and objective magnification.
Intelligent Inverted Fluorescence Microscope MIX60-FL
Adopting a modular design concept, the digital display LED fluorescence module enables three-color fluorescence observation, with the maximum customization of four-color fluorescence. It comes with an OLED digital display screen to intuitively show the current channel and light intensity. Each channel’s light intensity is independently memorized, eliminating the need for repeated adjustments when switching, making fluorescence intensity more precisely controllable—ideal for cellular fluorescence observation.

Microscope Camera MDX10
With 20-megapixel high resolution and a 1-inch large-format chip, it achieves 22 frames per second at full resolution, balancing high definition and speed.

High-Sensitivity Microscope Camera MSX3
Equipped with an innovative back-illuminated image sensor, it significantly enhances the digital camera’s performance under low-light conditions. The built-in ISP image processing chip is specially optimized for microscope shooting scenarios, accurately restoring the fine structure and true color of samples. It is an ideal tool for applications such as fluorescence photography, pathological diagnosis, and stereoscopic observation.

(2)Recommended MCS31/MCS11/21/22 for Full-Flow Monitoring and Analysis of Cell Culture Experiments
Live Cell Scanner MCS31
The MCS31 live cell scanner is an excellent partner for cell culture. It can be used inside an incubator, supporting phase-contrast and fluorescence observation of various culture flasks, dishes, 6-well plates to 384-well plates. It supports selected area scanning and stitching for large-field-of-view and long-term observation records of multi-well plates, greatly improving experimental efficiency in application scenarios such as comparative experiments and reducing the risk of experimental failure due to contamination.

Live Cell Imaging Systems MCS11/21/22
MSHOT live cell imaging systems MCS11/MCS21/MCS22 live cell imaging monitoring systems greatly simplify your workflow. Through automated timed observation, analysis, and reporting, you can remotely monitor cell culture progress without entering a cleanroom or opening the incubator—significantly improving work efficiency while avoiding the risk of interfering with growth and contaminating samples.


